Smart biosensor based on biofilm growth alteration as a warning system for aquatic pollution
The product consists in an innovative smart biofilm-based biosensor, design to detect biofilm electrochemical activity and proposed as a real-time biological early warning system (EWS) for in-field marine monitoring. In the first year of activity, we correlated changes in biofilm electrochemical activity with the presence of contaminants through experiments conducted under semi-field conditions (Figure 1). To assess the sensitivity, response time, and the potential application of this biosensor as a tool for evaluating anthropogenic impacts on the marine environment, the results were also compared with those obtained from standardized ecotoxicological bioassays (i.e. bacteria bioluminescence inhibition, algal growth inhibition, developmental anomalies).
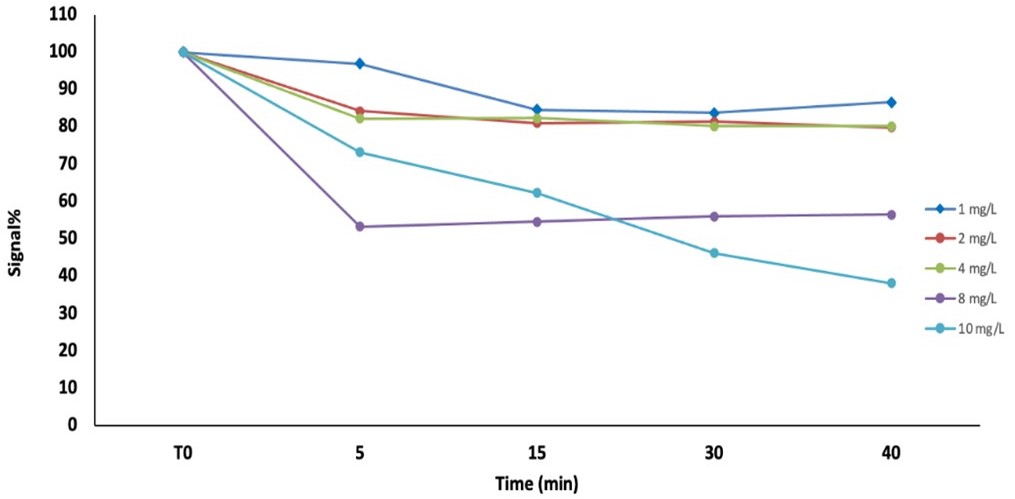
Figure 1. Biofilm Electrochemical Signal variation (%) after the exposure to different concentrations of fluoxetine (1, 2, 4, 8 and 10 mg/L) for 5, 15, 30 and 40 minutes.
The collected data demonstrated that this biofilm-based sensor can detect responses in seawater samples in a shorter time than traditional bioassays (5 minutes versus 30 minutes- 72h hours), confirming its potential as an effective early warning system for field applications. However, all tests were carried out using a commercial automated sensor originally designed to monitor biofilm formation in industrial process and production lines, an application designed to help preventing biofilm-related issues by supporting cleaning strategies. Given its promising potential, in the second year of the project we explored new insights and we introduced further improvements; specifically:
- monitoring of seasonal variations in biofilm growth and assessment of the influence of environmental factors (i.e., temperature, salinity, and pH) on biofilm formation dynamics;
- development of a new prototype equipped with a “multi-stem” system;
- experiments with independent “stems”, not connected to an electronic unit, to assess whether biofilm coverage reaches maturity and exhibits bio-electrochemical activity.
Influence of seasonal variations on biofilm growth
All experiments were conducted at the IAS-CNR Experimental Marine Station, located in Genoa harbour, between July 2024 and June 2025. The tests were carried out in a 100L tank with continuous renewal of natural seawater, pumped directly from the sea, where the biosensor (Figure 2) was continuously immersed to monitor variations in biofilm development. The system acquired the electric current signals (μA) generated by the electrochemical activity of the microbial biofilm in real time, at 3-second intervals, transmitting the data to a remotely accessible database. Once the electrochemical activity reached a plateau of approximately 30 μA – a value arbitrarily chosen to standardize the methodology (Gambardella et al. 2024) – the time required to reach this threshold was recorded. The sensor was then cleaned and re-immersed. All environmental parameters were regularly monitored on a weekly basis.
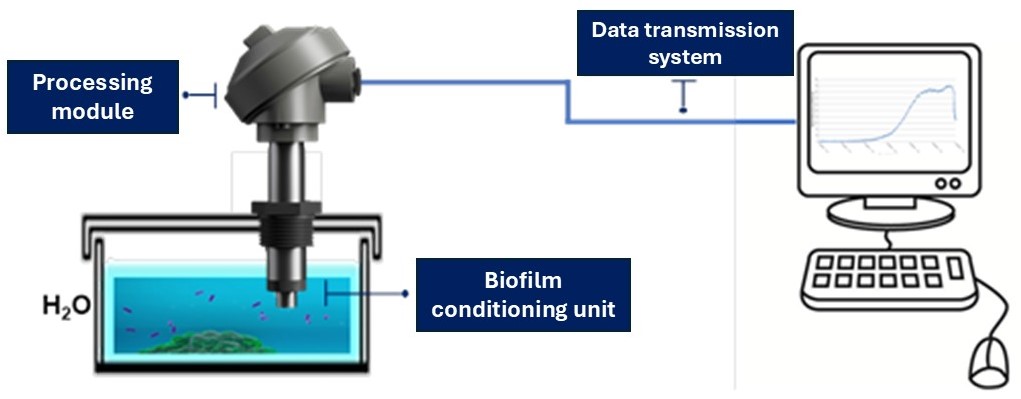
Figure 2. Main components of the biofilm-based biosensor: a biofilm conditioning unit, a signal transduction and processing module, and a data transmission system.
The data on environmental parameter monitoring are summarized in Table 1, while the results of biofilm growth monitoring are presented in Figure 3. Together, this information highlights which parameters are the most variable and limiting for biofilm growth in the marine environment. The graph shows that as temperatures decrease, the time required for biofilm development increases. Specifically, the biofilm reached the plateau value after an average of 5, 9, 16, and 12 days in summer, autumn, winter, and spring, respectively. The most limiting factors for biofilm growth were temperature, dissolved oxygen, and nutrient availability, while salinity and pH remained stable across seasons. Although low temperatures extended the bacterial growth lag phase, the high nutrient availability during winter and spring allowed the system to reach higher levels of bio-electrochemical activity. Overall, for laboratory experiments, the best compromise to ensure continuous sensor colonization by biofilm would be to maintain mesocosms at around 22°C, with continuous aeration and a high nutrient supply. Under these conditions, biofilm-coated sensors could be consistently available and could be used as early warning systems to monitor, identify, and prevent pollution in aquatic environments.
| Data | July 2024 | August 2024 | September 2024 | October 2024 | November 2024 | December 2024 | April 2025 | May 2025 | June 2025 |
|---|---|---|---|---|---|---|---|---|---|
| Salinity (PSU) | 37.02 | 37.41 | 37.17 | 36.58 | 37.04 | 36.02 | 36.01 | 36.2 | 36.6 |
| pH | 8.10 | 8.03 | 8.05 | 8.06 | 8.10 | 8.19 | 8.10 | 8.06 | 8.04 |
| Temperature (C°) | 27.94 | 29.11 | 23.5 | 20.85 | 18.96 | 12.85 | 23.1 | 25.5 | 26.3 |
| Dissolved 02 (ppm) | 5.51 | 4.92 | 4.04 | 6.19 | 7.12 | 8.86 | 6.5 | 7.71 | 5.7 |
| Biofilm growth (d) | 5.5 | 5 | 10 | 8.5 | 11 | 16 | 25 | 10 | 5.5 |
| Signal (µA) | 69 | 68.5 | 92 | 77.5 | 50 | 190 | 45 | 200 | 118 |
Table 1. Everage of environmental parameters (salinity, pH, temperature, dissolved oxygen) recorded during the tests, along with biofilm growth time and the corresponding bio-electrochemical activity signal.
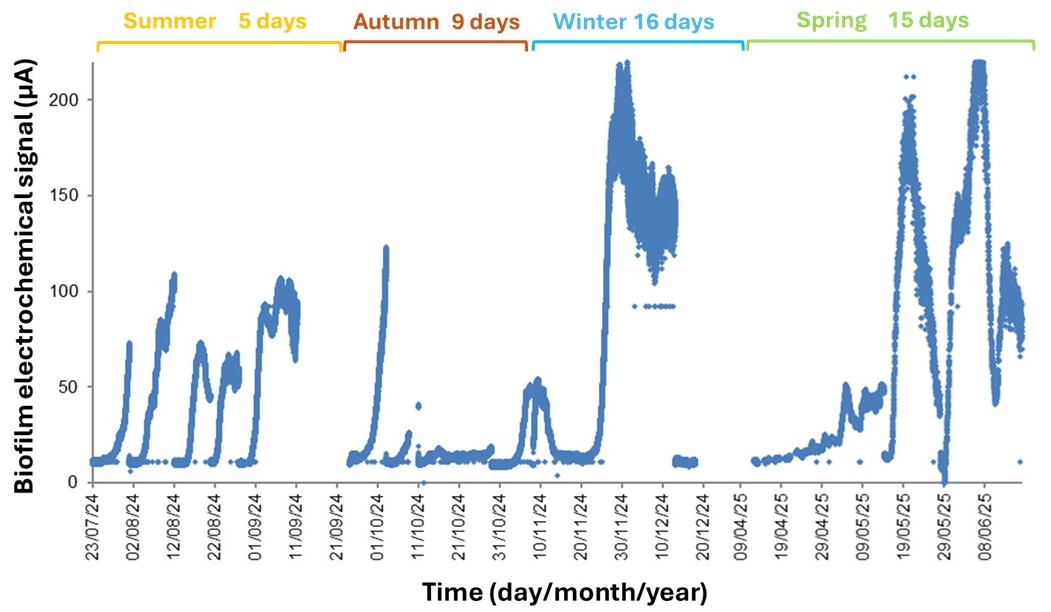
Figure 3. Representative electrochemical signal profile of biofilm development, highlighting seasonal-dependent variations during its formation. Specifically, biofilm develops after about 5, 9, 16 and 12 days in summer, autumn, winter, and spring, respectively. Each biofilm growth event was followed by cleaning of sensor and re-immersion.
Development of a new prototype equipped with a “multi-stem” system
Since the current sensor requires a new preconditioning period after each experiment—through cleaning and re-immersion until a new biofilm forms—a new prototype has been developed to overcome the limitation of a single “stem” biosensor. This advanced version integrates a single electronic unit with multiple “stems”, allowing the conditioning of different surfaces over time and facilitating the simultaneous execution of multiple tests within the same day (Figure 4).
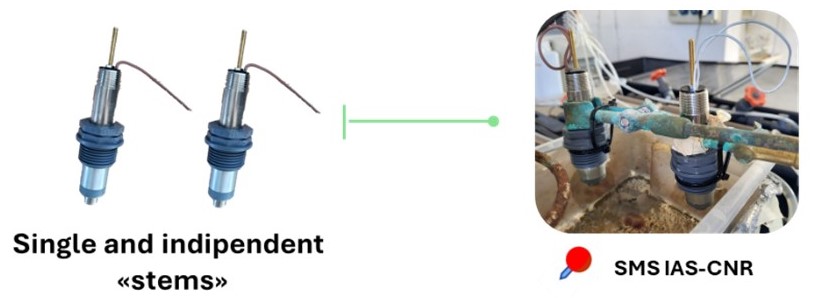
Figure 4. Example of the advanced version of the biofilm-based biosensor, which integrates a single electronic unit with multiple “stems”.
Laboratory experiments with multiple independent “stems”
Previous experiments showed that each “stem”, once connected to the electronic unit, was colonized by an electrochemical active biofilm within 5 to 16 days, depending on the season. However, additional laboratory experiments were carried out using independent “stems”, not connected to the electronic unit, in order to determine whether and when biofilm coverage had reached maturity and exhibited bio electrochemical activity. The possibility of immersing multiple “stems”, interchangeable with the electronic unit, would allow for a constant availability of bio-electroactive biofilm for in-field marine monitoring, without the need to clean the sensor and wait one or two weeks for recolonization each time. Initial tests were conducted by immersing different “stems”, not connected to the electronic unit, under both semi-field and laboratory conditions, and monitoring the biofilm’s electrochemical activity after one week. Specifically, the semi-field experiments were carried out at the IAS-CNR Experimental Marine Station, as previously described, while the laboratory tests were performed in a 125 L marine mesocosm, maintained under optimal conditions for biofilm growth (T = 22 °C, pH = 8, salinity = 36.6 ‰). Surprisingly, no variation in the bio-electrochemical activity (µA) was detected on the ‘stem’, suggesting that the presence of an electronic unit connected to the ‘stems’ may be essential for the development of an electroactive biofilm. To further investigate this issue, additional laboratory experiments were carried out by immersing titanium electrodes in the mesocosm under free corrosion conditions, where the metal corrodes naturally without any external electrical influence, and the corrosion rate is determined solely by the electrochemical balance between oxidation and reduction reactions.
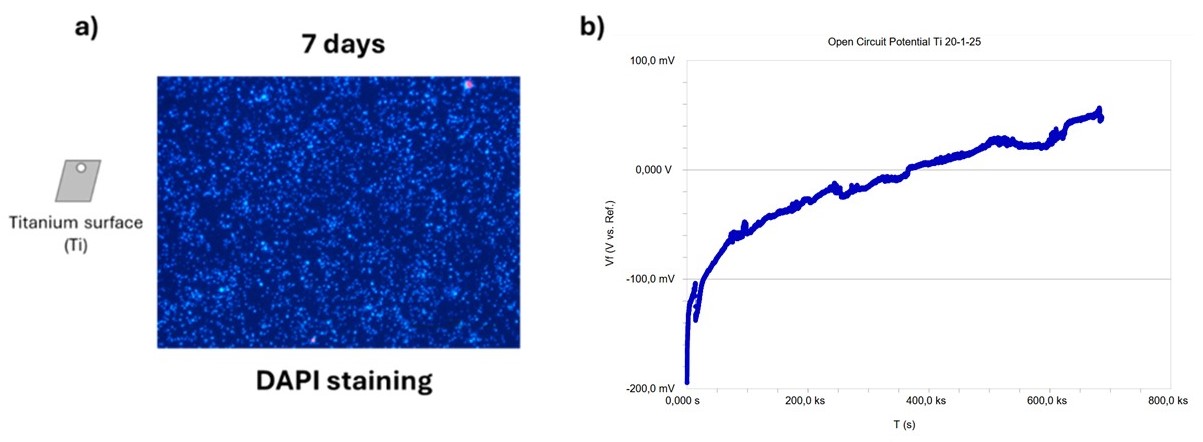
Figure 5. a) Biofilm colonization on titanium electrode revealed through DAPI staining; b) biofilm colonization on titanium electrode revealed through electrical potential monitoring.
The electrical potential was monitored daily to correlate its fluctuations with biofilm formation. In parallel, biofilm development on the titanium electrode was assessed using DAPI staining, a fluorescent dye that binds to DNA and enables visualization of bacterial colonization. As shown in Figure 5, DAPI staining revealed a high bacterial abundance on the titanium electrode, confirming the presence of biofilm. This finding is further supported by the electrical potential measurements, which showed an increase of 250 mV. These results demonstrate that, although the titanium sensor did not show an increase in electrochemical activity, it was colonized by biofilm. This led to the hypothesis that, in order to obtain a bio-electrochemical active biofilm, it is necessary to apply a constant potential to the series of “stems”. To this porpoise, further experiments were carried out by short-circuiting the zinc (part of the “stem” of the sensor) and titanium electrodes with a 20 kΩ resistor (Figure 6).
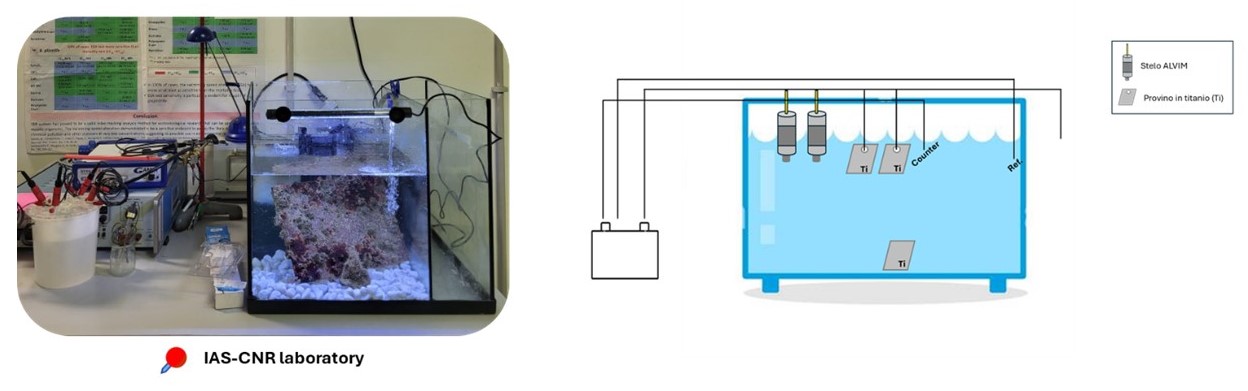
Figure 6. Schematic representation of the experiments carried out by short-circuiting the electrodes with a resistor.
As shown in Figure 7, DAPI staining confirms the presence of biofilm after 7 days of electrode immersion. In addition, the graph shows an increase in electric current of 20 µA from the beginning to the end of the test. Although this value is lower than the threshold previously chosen to standardize the methodology (30 µA), it indicates the onset of bio-electrochemical activity in the biofilm. In conclusion, to consistently obtain an electrochemically active biofilm, it is necessary to immerse multiple “stems” in series and apply a constant voltage.
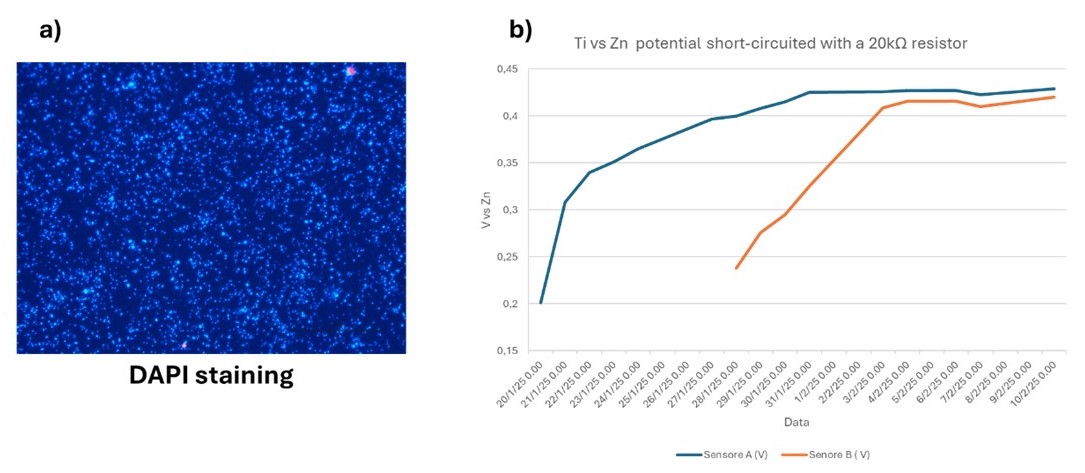
Figure 7. a) Biofilm colonization revealed through DAPI staining; b) biofilm electrochemical activity.
According to literature, electrochemical biosensors could provide rapid, sensitive, simple and low-cost measurements to assess overall toxicity in water samples by converting biological stress responses into quantifiable electrical signals (Gao et al. 2016; Hui et al. 2022). Our findings further indicate that this biofilm-based EWS offers several advantages, including i) real-time marine environment monitoring, ii) low maintenance requirements, iii) potential for serial assembly, iv) high stability, and v) low cost. These features make it particularly advantageous compared to molecule- and tissue-based biosensors, as biofilm forms spontaneously on the electrode surface and exhibits a longer operational lifespan, without the need for periodic renewal of the biological component. In addition, the possibility of assembling multiple sensors simultaneously ensures the continuous availability of biofilms in their “activated and responsive” phase, in accordance with seasonal temperature fluctuations. In conclusion, we highlight the potential of biofilm-based sensor for marine monitoring. However, long-term investigations are needed to validate these results and to evaluate the robustness, reliability, and accuracy of this innovative EWS under different environmental conditions and stressors, in order to support their potential integration into environmental monitoring programs at national and international level.

 Product 7.1
Product 7.1